Description
Buy Chlorphentermine Cas 461-78-9
Chlorphentermine, sold under the brand names
Apsedon,
Desopimon, and
Lucofen, is a
serotonergic appetite suppressant of the
amphetamine family. Developed in 1962, it is the
para-
chloro derivative of the better-known appetite suppressant
phentermine, which is still in current use.
The drug acts as a highly
selective serotonin releasing agent (SRA).
[4] It is not a
psychostimulant and has little or no
misuse potential, but is classed as a
Schedule III drug in the United States due mainly to its similarity to other appetite suppressants such as
diethylpropion which have been more widely misused. It is no longer used due mainly to
safety concerns, as it has a serotonergic effects profile similar to other withdrawn appetite suppressants such as
fenfluramine and
aminorex which were found to cause
pulmonary hypertension and
cardiac fibrosis following prolonged use.
[5]
Chlorphentermine was first
synthesized by 1954 and was subsequently developed in the early 1960s.
[6][7][8][9] It remained on the market in the United States as late as 2004.
[10]
Medical uses
Chlorphentermine was used as an
appetite suppressant for purposes of
weight loss in people with
overweightness or
obesity.
[11][3][12]
Side effects
Side effects of chlorphentermine include
impaired sleep,
irritability, and
gastrointestinal symptoms including
dyspepsia.
[11]
Euphoria is said to occasionally occur with chlorphentermine, but to a much lesser extent than with
dextroamphetamine.
[3] The drug does not produce
amphetamine-like subjective effects in humans
[13][14] and the
psychostimulant effects of chlorphentermine are described as much less than those of dextroamphetamine.
[3]
Pharmacology
Pharmacodynamics
Chlorphentermine acts as a
selective serotonin releasing agent (SSRA).
[25][26][18] The
EC50Tooltip half-maximal effective concentration for monoamine release with chlorphentermine are 30.9
nM for
serotonin, >10,000
nM for
norepinephrine, and 2,650
nM for
dopamine.
[25][26][18] Although it is inactive as a norepinephrine releasing agent, it is a moderately
potent norepinephrine reuptake inhibitor (
IC50Tooltip half-maximal inhibitory concentration = 451
nM; 15-fold lower than its
EC50 value for serotonin release).
[27][26] The activity of chlorphentermine as an SSRA is in contrast to
phentermine, which acts as a selective
norepinephrine and dopamine releasing agent (NDRA).
[26][18]
In animals, chlorphentermine robustly and
dose-dependently increases
serotonin levels in the brain.
[23][14] It also increases
dopamine levels in the brain at high doses.
[23][14] Whereas
dextroamphetamine and
phentermine robustly
stimulate locomotor activity and are
self-administered in animals, chlorphentermine does not increase locomotor activity and is either not self-administered or is only weakly self-administered.
[28][13][14] Conversely, it has been reported that chlorphentermine weakly stimulates locomotor activity at low doses and progressively suppresses it at higher doses.
[29] In contrast to other amphetamines, it does not produce
stereotypies nor reverse
reserpine-induced behavioral depression.
[29] In addition, unlike
para-chloroamphetamine (PCA), chlorphentermine does not produce the
head-twitch response, a behavioral proxy of
psychedelic effects, in animals.
[29]
In contrast to
fenfluramine and
norfenfluramine, chlorphentermine shows negligible activity as an
agonist of the
serotonin 5-HT2A,
5-HT2B, and
5-HT2C receptors.
[26] Its
EC50 values at these receptors are >10,000
nM, 5,370
nM, and 6,456
nM, respectively.
[26] These
EC50 values are >324-fold, 164-fold, and 209-fold lower than its
EC50 value in inducing serotonin release, respectively.
[26]
Despite its lack of direct agonism at the serotonin 5-HT
2B receptors, chlorphentermine shows induction of
primary pulmonary hypertension in
animal models.
[26][3] This suggests that serotonin release can induce this form of toxicity without concomitant direct serotonin 5-HT
2B receptor agonism.
[26] However, other findings seem to contradict this hypothesis, for instance increases in serotonin levels with fenfluramine and other serotonin-elevating drugs being inadequate for inducing cardiac valvulopathy-like changes, and instead implicating additional direct serotonin 5-HT
2B receptor agonism in this toxicity.
[30] It has been said that it is possible that an
active metabolite of chlorphentermine might show greater serotonin 5-HT
2B receptor agonism than chlorphentermine itself, analogously to the case of fenfluramine and
norfenfluramine, and that this possibility should be examined.
[26]
The amphetamine homologue of chlorphentermine, PCA, is a
potent serotonergic neurotoxin.
[31] In contrast to PCA, preliminary animal experiments suggest that chlorphentermine is non-neurotoxic, although more research in this area is still needed.
[31]
Pharmacokinetics
The
elimination half-life of chlorphentermine is relatively long and is stated to be 40
hours
[2] and about 5
days by different sources.
[3]
Chemistry
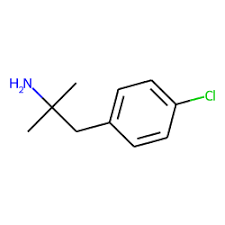
Chlorphentermine, also known as 4-chlorophentermine, 4-chloro-α-methylamphetamine, and 4-chloro-α,α-dimethylphenethylamine, is a
substituted phenethylamine and
amphetamine derivative.
[23] It is the
para-
chloro analogue of
phentermine.
[23] Chlorphentermine is also closely structurally related to certain other phentermines including
cericlamine,
cloforex,
clortermine,
etolorex, and
methylenedioxyphentermine (MDPH). It is closely structurally related to the amphetamine derivatives
para-chloroamphetamine (PCA) and
para-chloromethamphetamine (PCMA) as well.
[32]
History
Chlorphentermine was first described in the
scientific literature by 1954.
[6][7][8][9] It was subsequently developed for use as an appetite suppressant in the early 1960s.
[6][7][8][9] The drug is said to have been
withdrawn from the market in the United States in 1969
[33] and in the United Kingdom in 1974.
[3] However, other sources indicate that chlorphentermine continued to be marketed in the United States as late as 2004.
[10][34] Pulmonary toxicity of chlorphentermine was observed in animals by the early 1970s and this resulted in reservations about its clinical use.
[3]
Chlorphentermine supplier | CAS 461-78-9
AOBIOUS
https://aobious.com › aobious › custom-synthesis › 241...
view and buy high purity Chlorphentermine from AOBIOUS, the leading supplier of life science reagents.
Chlorphentermine Hydrochloride
LGC Standards
https://www.lgcstandards.com › TRC-C375080
Buy Chlorphentermine Hydrochloride online at LGC Standards. High quality reference ... Alternate
CAS Number. Free base:
461-78-9. Drug Type. Amphetamines ...
Read more
Chlorpheniramine . maleate - Enzo
enzo.com
https://www.enzo.com › Shop
CAS. 113-92-8. Formula. C16H19ClN2 C4H4O4. MW. 390.87. Purity. ≥98%. Solubility. Soluble in water (250 mg/mL) and ethanol (100 mg/mL).
Read more
Chlorphentermine hydrochloride
marquessilvaneves.com
https://marquessilvaneves.com › ... › Reference Standards
Chlorphentermine hydrochloride is a serotonin transporter (SERT) substrate with a Ki value of 338 nM for [3H]5-HT uptake.
Read more
Chlorphenamine maleate EP Reference Standard CAS ...
Sigma-Aldrich
https://www.sigmaaldrich.com › sial
Buy Chlorphenamine maleate Ph Eur reference standard for identification, purity tests or assays of pharmaceutical products according to EP monographs.
LGD-3303 [CAS: 917891-35-1] Glixxlabs.com High Quality ...
glixxlabs
https://www.glixxlabs.com › GLXC-15904
Novel selective androgen receptor modulator (SARM), acting as a partial agonist for androgenic effects, but a full agonist for anabolic effects.
Read more
Chlorpheniramine Maleate (CAS 113-92-8)
Santa Cruz Biotechnology
https://www.scbt.com › chlorpheni...
Chlorpheniramine Maleate est un antagoniste du récepteur H1 de l'histamine. Numéro CAS: 113-92-8. Pureté: ≥99%. Masse Moléculaire:.
Read more
chlorоphentermine CAS#: 461-78-9
ChemicalBook
https://www.chemicalbook.com › ProductChemicalProp...
Chlorphentermine, a sympathomimetic phenethylamine derivative,
was introduced over twenty years ago for the treatment of obesity. Concern that its use was ...
Read more
NIDA Drug Supply Program Catalog (2019)
National Institute on Drug Abuse (NIDA) (.gov)
https://nida.nih.gov › files › ndsp_catalog_29
Name :
Chlorphentermine; 4-Chloro-α,α-dimethyl-β-phenethylamine. Mol. formula : Merck Index, 14th ed., Monograph 2182. Anorectic. CASRN :
461-78-9. Notes ...
Read more212 pages
Chemical Properties of Chlorphentermine (CAS 461-78-9)
Cheméo
https://www.chemeo.com › cid › Chlorphentermine
Reviews
There are no reviews yet.